An Electron's Position Is Best Described by Probability
It has to do with. The Heisenberg uncertainty principle states that the exact position and momentum of an electron cannot be simultaneously determined.
The Quantum Mechanical Model Of The Atom Article Khan Academy
Lithium Li contains three electrons that occupy the first and second.
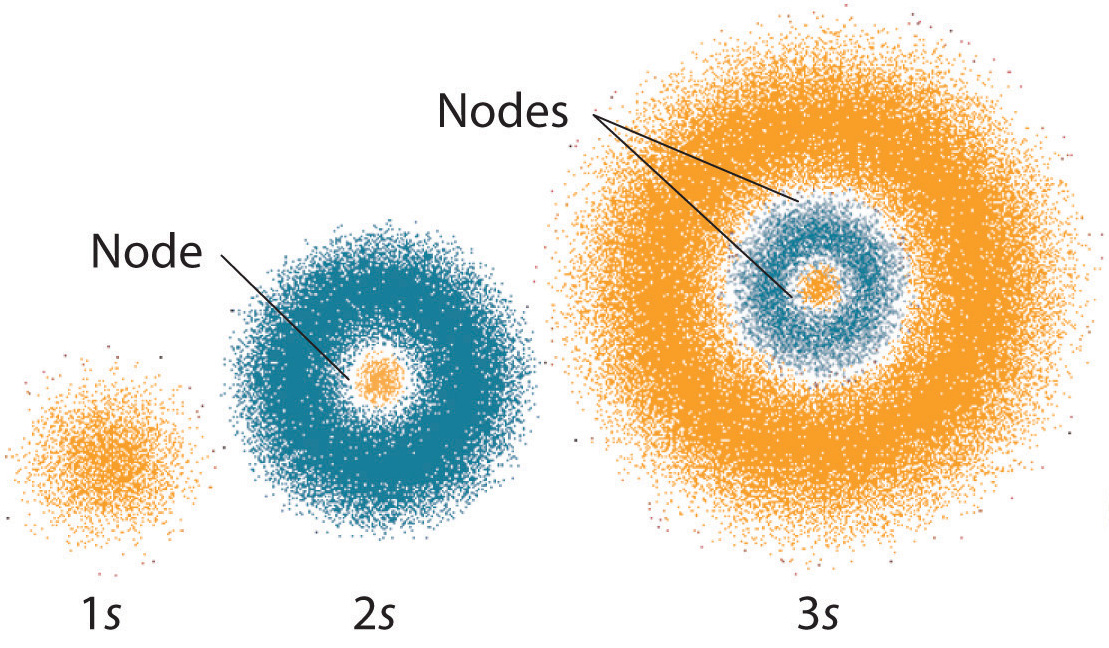
. As has been stated you let an electron hit a screen that will leave a mark or a signal. Compute the expectation value of the. The 1 s orbital is spherically symmetrical so the probability of finding a 1 s electron at any given point depends only on its distance from the nucleus.
Once each p orbital has an electron a second may be added. 1 If the electrons position can be measured to an accuracy of 20x10-8 m how accurately can its speed velocity be known. When filling the p orbitals each takes a single electron.
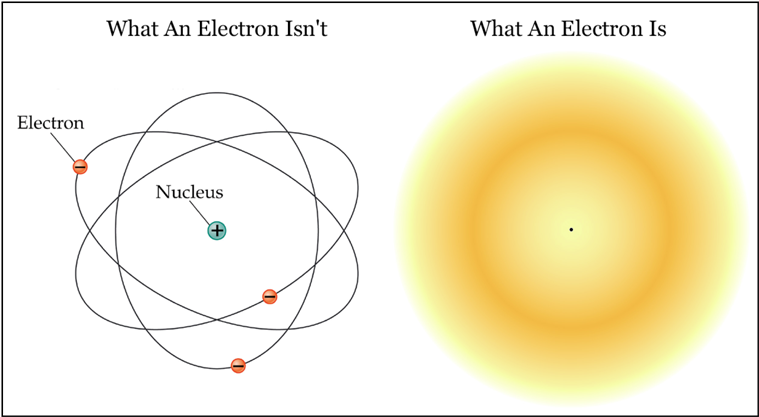
Previous models described the motion of electrons in the same was as the motion of large objects. For an electron in a hydrogen atom at the ground electronic state Is orbital what is the probability that a measurement of the electrons position will yield a value r 2a. In quantum mechanics an electron doesnt have a definite position or momentum.
Physics questions and answers. A three-dimensional region around the nucleus of an atom that describes an electrons probable location. Two important refinements resulting from the wave-mechanical atomic model are 1 that electron position is described in terms of a probability distribution and 2 electron.
Now it is general agreed that we can only speak of probability -- the probability of finding an. It has a wave function from which the probability of finding it at a particular position or momentum can be calculated. Daltons model of an atom is best described as a what.
The possible energies that. Suppose an electron is in a state described by the wave function 0 gr e1 sin cos V4T where I gr 2 2 dr 1 and 0 are azimuthal and polar angles respectively. The lowest orbit has two electrons 1s2 the next orbit has eight 2s2 2p6 etc.
The theory of quantum mechanics says that an electrons position can only be described statistically. I can also make a very small slit and if I detect a signal after the slit I know that the. The probabilities on an electrons position are best visualized with an electron _____ diagram.
An electron bound to a proton will probably be very near the proton. Who was the first to provide evidence that atoms contain subatomic particles. Sommerfeld worked out the shapes and probabilities of location and speed of electrons in the.
The probability density is greatest at r 0. The quantum mechanical model of the atoms estimated the probability of finding. A An electron in a carbon nanotube of length L is described by the wavefunction ψ x sin 2 π x L psi x sin 2 pi x L ψ x sin 2 π x L.
We can think of a spherical cloud around the nucleus that is darker near the center and grows paler as the distance from the center increases. For the electrons position in a 1s orbital the probability density rhor is maximal at the origin whereas the radial probability density pr is maximal at the Bohr. And a What are the possible.
Electron Densityor Probability Density. Position is described in terms of a probability distribution and 2 electron energy is quantized into both shells and subshells--each electron is characterized by four quantum numbers. It also describes a method for calculating the probability of finding an electron at.

2 10 Quantum Mechanics And The Atom Chemistry Libretexts

Ask Ethan What S The Difference Between A Fermion And A Boson Wave Function Quantum Mechanics Quantum Physics
Comments
Post a Comment